EspeRare has entered into a partnership with the Pierre Fabre group to develop ER004, a novel in utero protein replacement therapy for the treatment of X-Linked Hypohidrotic Ectodermal Dysplasia (XLHED), a rare pediatric genetic disease.
There is currently no approved therapy for the treatment of XLHED and ER004 has the potential to become the first ever prenatally administered drug to correct a genetic disease before birth. During the second half of 2021 and in view of discussions with the EU and US regulatory agencies, both partners aim to start enrolling patients into a clinical trial geared towards marketing approval.
With Pierre Fabre, EspeRare is fortunate to have found a partner that shares common values and patient-centered commitments.
Under the terms of the agreement, EspeRare and Pierre Fabre will become co-sponsors of the development of ER004 in Europe and in the US. Pierre Fabre will be responsible for its commercialization worldwide. In exchange for exclusive commercial rights, EspeRare is receiving financial returns. Delivering on EspeRare non-profit model, these financial returns are on one hand, shared with past contributors of ER004 development and on the other hand, reinvested into scaling EspeRare’s organization and portfolio of therapeutic programs.
Eric Ducournau, CEO of the Pierre Fabre Group commented: « We are proud to ally with the EspeRare foundation to find a therapeutic solution to a debilitating disease affecting 200 babies in Europe every year. We cannot think of a better way to epitomize our corporate purpose: “Every time we take care of one single person, we make the world better »
Caroline Kant, Founder and CEO of EspeRare, stated: “At Esperare we are thrilled to collaborate with the Pierre Fabre Group to co-develop this very inspiring program. We are confident that joining forces with Pierre Fabre will allow to bring this ground-breaking therapy to XLHED patients in alignment with our patient-centered values.”
Presses releases
Please find the full press release in English and in French.
XLHED is a severe, chronically debilitating and life-threatening rare disease affecting approximately 4/100,000 live male births every year. XLHED is caused by genetic mutations in the EDA gene, a gene that encodes for an important ectodermal developmental protein, EDA. The absence of functional EDA results in abnormal development of the skin, sweat glands, sebaceous glands, hair, oral cavity, and respiratory mucosal glands resulting in serious life-threatening clinical manifestations from birth including hyperthermia, craniofacial anomalies and recurrent respiratory infections that impair quality of life in patients and their families.
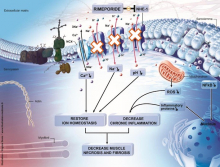
ER004 is a fully humanized EDA molecule consisting of the human IgG1 Fc sequence linked to the human EDA TNF binding domain. Preclinically, ER004 has been shown to bind to the receptor EDAR resulting in activation of the NFϰB signaling pathway, which triggers the transcription of genes involved in the normal development of multiple tissue types. ER004 is the first and only treatment specifically targeting XLHED. Administered during the third trimester of pregnancy, it has the potential to become a “single course” treatment, significantly improving symptoms of the disease throughout patients’ lives. This approach has already demonstrated significant potential in a case series of three patients treated in utero with ER004 during the third trimester of pregnancy. The treatment normalized sweat gland function and associated thermoregulation, and improvement in dentition and respiratory function were observed. These results were recently published in the New England Journal of Medicine and featured in Nature Medicine’s 2018 Research Highlights.
Pierre Fabre is the 2nd largest private French pharmaceutical group and 2nd largest dermo-cosmetics laboratory in the world. Its portfolio is made of medical franchises (oncology, dermatology, pharmaceutical care) and leading dermo-cosmetic brands (Eau Thermale Avène, Ducray, A-Derma, Klorane, René Furterer, Pierre Fabre Oral Care…). In 2019, Pierre Fabre generated revenues of €2.4 billion, two-thirds of which came from international sales. Headquartered in the South-West of France, the Group employs some 10,400 people worldwide, owns subsidiaries or offices in 45 countries, and distributes its products in over 130 countries.86% of the Pierre Fabre group is held by the Pierre Fabre Foundation, a government-recognized public-interest foundation, while a smaller share is owned by its employees via an employee stock ownership plan.In 2019, Ecocert Environment assessed the Group’s CSR approach in accordance with the ISO 26000 sustainable development standard and awarded it the Ecocert 26000 “Excellence” rating.For more information, please visit www.pierre-fabre.com